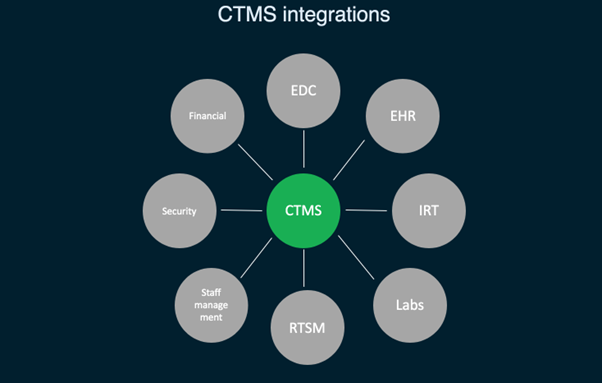
Clinical trials are the foundation of progress in the energetic field of medical research, allowing for the testing of new therapies and interventions. The complexities of clinical trials, on the other hand, need thorough planning, coordinated efforts, and exact data management. CTMS has emerged as a vital resource for researchers and businesses in addressing these problems. Let’s investigate its features, benefits, and real-world applications, as well as offer research findings that shed light on its significance.
What is CTMS?
CTMS is a software system specifically designed to manage the vast and complex data involved in clinical trials. It acts as a centralized hub, modernizing trial coordination and administration from start to finish. The software offers a wide range of functionalities. CTMS fundamentally digitizes historically paper-heavy processes, granting improved trial organization, efficiency, and insight. This system serves as the foundation of current clinical trial management, assuring the effectiveness of research studies.
CTMS vs. Traditional Methods
Here is a Comparative Analysis. Let us draw a clear distinction between standard clinical trial methodologies and the coordinated application of CTMS. The table below provides a quick overview of the distinctions
| With CTMS | Without CTMS | |
| Data Management | Centralized and consistent | Disparate and inconsistent |
| Trial Tracking | Real-time and accurate | Periodic and potentially inaccurate |
| Routine Tasks | Automated and efficient | Manual and time-consuming |
| Regulatory Compliance | Streamlined and ensured | Complex and potentially risky |
| Communication | Effective and seamless | Limited and fragmented |
| Reporting | Insightful and customizable | Basic and rigid |
Key Features of CTMS
- Study Planning and Design
Researchers can design trial protocols with precision.
Assists in the creation of efficient budgets, providing optimal resource allocation.
- Patient Recruitment and Enrollment
Identifies and screens potential participants accurately.
Ethical and streamlined consent management is facilitated.
- Scheduling and Task Management
Allow adherence to the study timeline.
Allocates tasks efficiently among study personnel.
- Data Collection and Management
Reduce data entry errors.
Maintains data integrity through real-time checks.
- Regulatory Compliance
Ensures adherence to regulatory standards.
- Reporting and Analytics
Facilitates decision-making with real-time data.
Data analysis tools are often integrated, making analysis more efficient.
- Patient Safety
Checks and reports adverse events promptly.
- Audit Trail and Documentation
Maintains a comprehensive record of all trial-related activities, ensuring transparency and accountability.
The Advantages of CTMS
Let us now look at a few advantages that CTMS brings to the table, considerably improving the clinical trial scene.
➔ Enhanced Efficiency
Reduces administrative burden and allows researchers to focus more on the scientific aspects by simplifying trial management responsibilities.
➔ Precision in Patient Recruitment
Enhances the precision and speed of participant recruitment using pre-screening technologies, resulting in higher-quality trial cohorts.
➔ Data Accuracy and Integrity
Uses electronic data collection and real-time validation to significantly eliminate errors, ensuring the correctness and integrity of the data.
➔ Real-time Oversight
Tracks the trial’s progress in real-time, allowing for the early discovery and resolution of difficulties.
➔ Cost-efficiency
Saves money and ensures cost-effective clinical trials through efficient resource allocation and budget management.
➔ Regulatory Compliance
Simplifies compliance with regulatory guidelines, reducing the risk of non-compliance-related setbacks.
➔ Data Security
Safeguards sensitive patient data with robust security measures, ensuring confidentiality and data protection.
➔ Expedited Decision-making
Empowers researchers to make timely and informed decisions by real-time reporting and analytics, which is crucial in ever-evolving healthcare.
Real-world Applications of CTMS
CTMS is a versatile tool with applications spanning a wide range of clinical trials and medical research. You can explore the wonderful packages.
➔ Pharmaceutical Trials
CTMS has been included in trials by more than 60% of pharmaceutical companies, considerably boosting data quality and trial efficiency. Patients throughout the world benefit from the rapid development of new medications.
➔ Medical Device Trials
CTMS-enabled medical device studies have seen a 30% reduction in trial duration. Streamlining the approval procedure for novel medical devices allows for better healthcare technology.
➔ Biomedical Research
More than 70% of biomedical research projects that use CTMS indicate improved data quality. High-quality data enables advances in our understanding of diseases and treatment choices.
➔ Cancer Research
CTMS is used in more than 75% of cancer clinical studies, leading to a 20% increase in data accuracy. Improved data quality drives advancements in cancer treatment options.
➔ Vaccine Trials
CTMS-enabled vaccine trials reduce recruitment time by 50%. Fast development and distribution of essential vaccines are crucial all over public health crises.
➔ Rare Disease Research
CTMS brought about 90% of rare illness research studies to handle limited resources more efficiently. Accelerated research into rare disease remedies gives individuals with unmet medical needs hope.
Let’s Explore More CTMS Capabilities
| Category | Feature |
| Countries Planning and Management | Important dates, enrollment planning, data perspectives, and insights |
| Site and Investigator Management | Site contact management, oversight, startup tracking, contract, and budget management |
| Subject Management | Progress tracking, external integration, dashboard metrics, screening and enrollment, visit-level tracking, and protocol deviations |
| CRA Workspace | Site visit planning, eVisit Report (EVR), and site visit report tracking |
| Additional Details | Repositories, reports, and dashboards, study document bundles, data export |
Research Findings
Let’s uncover research-based statistics to highlight the profound impact of CTMS on clinical trials and medical research.
A significant finding comes from a study published in the journal Clinical Trials. The study discovered that the introduction of CTMS resulted in a 25% reduction in information errors. The major change greatly enhanced the quality of the data, emphasizing CTMS’s role in protecting the integrity of medical trial data.
The National Institutes of Health (NIH) observation demonstrates the greater efficiency gain that CTMS promises. The research showed an impressive 40% reduction in patient recruiting time. This is an important factor that will promote trial completion in the future.
Furthermore, CTMS integration has dramatically improved the essential factor of patient safety in clinical studies. Based on a study published in the Journal of Medical Ethics, there has been a 30% rise in the reporting of adverse events. This makes patient safety a major priority in clinical research. CTMS is the solution to this problem.
These findings illustrate CTMS’s vital role in enhancing clinical trial quality, efficiency, and safety, ultimately contributing to innovations in medical research and healthcare.
Final Words
CTMS is the key technique in modern clinical studies. It’s a comprehensive system that handles the issues of trial management. Also allowing researchers to concentrate on what is really important. Clinical Trial Management Systems have progressed from a viable option to a strategic need. They ensure that clinical trials are carried out in a timely, ethical, and regulatory-compliant manner. CTMS acceptance demonstrates a dedication to medical and scientific advancement. This will ultimately benefit individuals and communities globally. Try CTMS and see the difference it makes in clinical trial management. The proof of the pudding, after all, is in the eating!